Great American Assisted Living Community at Bradenton, LLC is a for-profit ALF located at 1015 7th Avenue East, Bradenton, Florida 34208 with 135 licensed beds. This limited liability company took over operations in late 2022.
Since then, investigators with the Florida Agency for Health Care Administration have inspected the facility and issued citations of deficiencies based on findings that it failed to comply with Florida law.
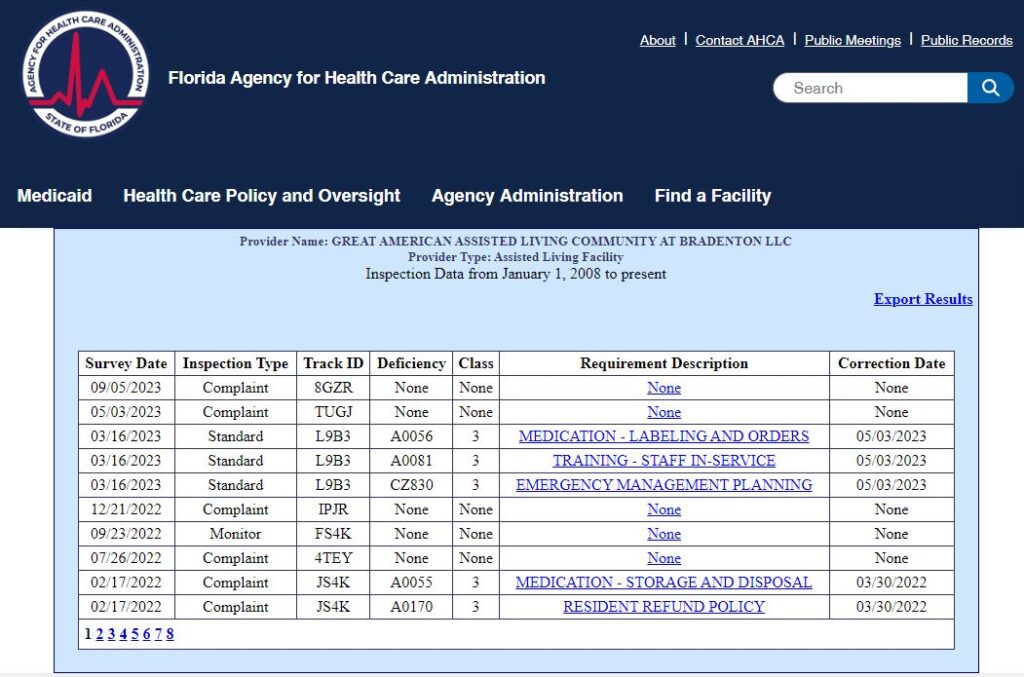
Great American Assisted Living Community at Bradenton Cited with Deficiencies

Great American Assisted Living Community at Bradenton was cited with a total of three Class III deficiencies during 2023. These deficiencies were related to the failure to follow requirements for medication labeling and orders, staff in-service training and emergency management planning. For the life, health and safety of its residents, the handling, labeling, ordering and assistance with medications in an assisted living facility is of considerable importance.
The legal requirement for medication labeling and orders provides that: MEDICATION LABELING AND ORDERS. (a) The facility may not store prescription drugs for self-administration, assistance with self-administration, or administration unless they are properly labeled and dispensed in accordance with Chapters 465 and 499, F.S., and Rule 64B16-28.108, F.A.C. If a customized patient medication package is prepared for a resident, and separated into individual medicinal drug containers, then the following information must be recorded on each individual container: 1. The resident’s name; and, 2. The identification of each medicinal drug in the container. (b) Except with respect to the use of pill organizers as described in subsection (2), no individual other than a pharmacist may transfer medications from one storage container to another. (c) If the directions for use are “as needed” or “as directed,” the health care provider must be contacted and requested to provide revised instructions. For an “as needed” prescription, the circumstances under which it would be appropriate for the resident to request the medication and any limitations must be specified; for example, “as needed for pain, not to exceed 4 tablets per day.”
The revised instructions, including the date they were obtained from the health care provider and the signature of the staff who obtained them, must be noted in the medication record, or a revised label must be obtained from the pharmacist. (d) Any change in directions for use of a medication that the facility is administering or providing assistance with self-administration must be accompanied by a written, faxes, or electronic copy of a medication order issued and signed by the resident’s health care provider. The new directions must promptly be recorded in the resident’s medication observation record. The facility may then obtain a revised label from the pharmacist or place an “alert” label on the medication container that directs staff to examine the revised directions for use in the medication observation record. (e) A nurse may take a medication order by telephone. Such order must be promptly documented in the resident’s medication observation record.
The facility must obtain a written medication order from the health care provider within 10 working days. A faxed or electronic copy of a signed order is acceptable. (f) The facility must make every reasonable effort to ensure that prescriptions for residents who receive assistance with self-administration of medication or medication administration are filled or refilled in a timely manner. (g) Pursuant to Section 465.0276(5), F.S., and Rule 61N-1.006, F.A.C., sample or complimentary prescription drugs that are dispensed by a health care provider, must be kept in their original manufacturer’s packaging, which must include the practitioner’s name, the resident’s name for whom they were dispensed, and the date they were dispensed. If the sample or complimentary prescription drugs are not dispensed in the manufacturer’s labeled package, they must be kept in a container that bears a label containing the following: 1. Practitioner’s name, 2. Resident’s name, 3. Date dispensed, 4. Name and strength of the drug, 5. Directions for use; and, 6. Expiration date. (h) Pursuant to Section 465.0276(2)(c), F.S., before dispensing any sample or complimentary prescription drug, the resident’s health care provider must provide the resident with a written prescription, or a faxed or electronic copy of such order.
Additionally, complaints that were filed against the facility with AHCA were investigated by the Agency and reported on in May and September of 2023. No deficiencies were issued in connection with these complaints.
The complete inspection report and history of complaints and deficiencies for this Bradenton assisted living facility may be viewed here http://tinyurl.com/2p9jpybn
Do you need to discuss an ALF issue or complaint with an Elder Abuse Lawyer? Call (941) 485-7600

If you have questions or concerns about the safety of a loved one in a Florida assisted living facility, you may call and speak with directly. I provide a free, confidential consultation at (941) 485-7600. James Keim, Florida Assisted Living Facility Neglect and Abuse Lawyer serving Bradenton, Sarasota and surrounding Florida communities.
By holding assisted living facilities accountable for the severe harm they can cause to our seniors, we can fight to improve the quality of care for all Floridians. For more information, visit https://venicelawfirm.com/
File under: Great American Assisted Living reviews complaints lawsuits
File under: Google reviews, complaints, Bradenton assisted living facility reviews and complaints and investigations.